About the Report
A growing number of biologic medicines have been developed and approved over the past decade, improving the lives of patients worldwide. However, patient access to biologics has been limited, partly due to their relatively high cost. Biosimilars are increasingly available across Europe, bringing with them the opportunity to generate competition for biologic therapies and thereby lower medicine costs and increase patient access. This report puts forth a framework for the evaluation of sustainability in the biosimilars market and identifies its key policy elements. Based on these criteria, current policies and market dynamics are assessed to identify where risks to sustainability exist and the best practices that can support the long-term positive contribution of biosimilars in Europe.
Report Summary
Biosimilars are now an integral part of the market for biologics, which accounted for $277 billion in sales globally in 2017 and is projected to reach $452 billion by 2022. Use of biosimilars in the EU5 countries alone stands to offer savings of more than €10 billion between 2016 and 2020.
To sustain the market for biosimilars in the long-term, ongoing benefits for all stakeholders must be ensured. Sustainable policies therefore provide for patient access and physician prescription choice, provide a means to manage existing healthcare budgets for payers, and safeguard a healthy level of competition, supply, and product safety and quality.
Current European policies and market dynamics were assessed to identify the risks and challenges to sustainability, with the aim to identify best practices that can be leveraged to support long-term sustainability of the biosimilars market in Europe. Overall, biosimilars have increased patient access to biologic medicines, and this growth is supported by a regulatory environment and clinical guidelines that create a neutral or positive climate for biosimilars relative to originator biologics, and thus supports their sustainability.
However, some elements of a sustainable system were found to potentially be at future risk due to payer driven-switch and specific purchasing systems. Specifically, payer-driven switch was found to reduce physician prescription choice, potentially leading to loss of product options currently working for patients and reducing patient involvement in the treatment decision. Additionally, single-winner tenders were found to disrupt market forces and competition, thereby not supporting long-term sustainability. Several alternative policies currently in place in Europe can be leveraged to ensure and support the long-term sustainability of the biosimilars market while additionally fulfilling the needs of all stakeholders.
Key Findings
Sustainability in the biosimilars marketplace ensures benefits for all stakeholders

- Biosimilars are a key means to alleviate financial challenges faced by many stakeholders in the currently constrained European budgetary environment.
- Elements of biosimilar sustainability represent the needs of all stakeholders including patients, physicians, payers and manufacturers and support the healthy functioning of the marketplace for medicines.
- Policies can influence the achievement of these elements variably, and it is therefore valuable to explore which current biosimilar policies in Europe best secure a sustainable environment supporting these.
Launch of a biosimilar brings with it increased patient use of biologic medicines
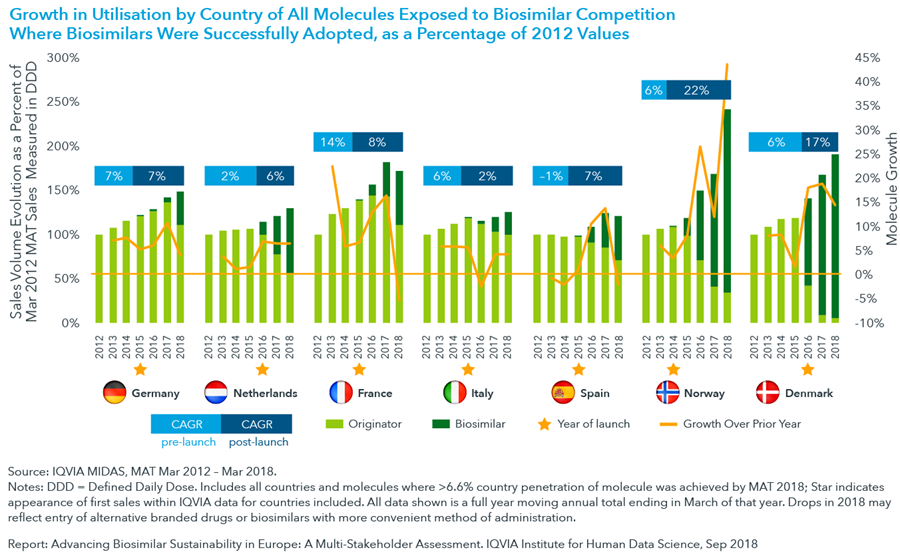
- In the years following biosimilar launch, significant growth is seen in the use of biologic molecules. However, growth often slows to pre-launch levels within a few years, reflecting the declining competitiveness of the molecule itself within a market that includes innovative therapies.
- Uptake also varies by country; in Norway, where biosimilars are strongly promoted by health authorities, and national tenders are used, uptake is fast and use of biologic medicines grow. In contrast, in countries like Italy, where health authorities have a more conservative perspective regarding biosimilars, the uptake has been slower.
- Faster and higher biosimilar penetration is seen when national tenders are run as single tenders with single winners, as in the case of Norway and Denmark, however this mechanism was found to jeopardise long-term sustainability.
The European regulatory environment and clinical guidelines are generally positive toward sustainability
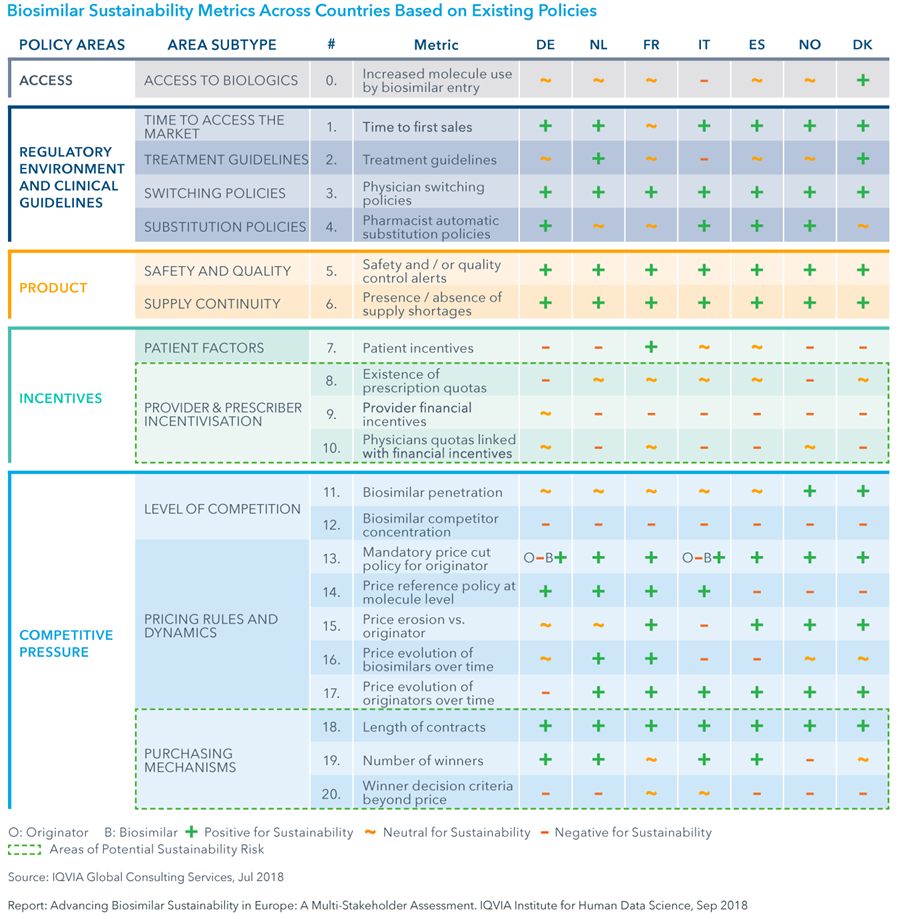
- Overall, the current European regulatory environment creates a neutral or positive environment for biosimilars relative to originator biologics and supports sustainability.
- However, two potential areas pose a risk to sustainability: payer-driven switch and tender systems that award total molecule market volume to a single competitor.
- Payer-driven switch was found to reduce physician prescription choice, potentially leading to loss of product options currently working for patients and to reduce patient involvement in the treatment decision.
- Although single-winner tenders were found to achieve greatest price reduction on biologic products when biosimilar competition exists, they were also found not to support long-term sustainability as they disrupt market forces and competition by excluding nonwinner manufacturers from the market for the duration of the tender contract.
- Additional evidence suggests single-winner tenders do not always optimise savings, since physicians can still use non-preferred product at a higher price; whereas multi-winner tenders offer price reductions on all contracted products.
Several policies currently in place in Europe can be leveraged to support the long-term sustainability of the biosimilars market
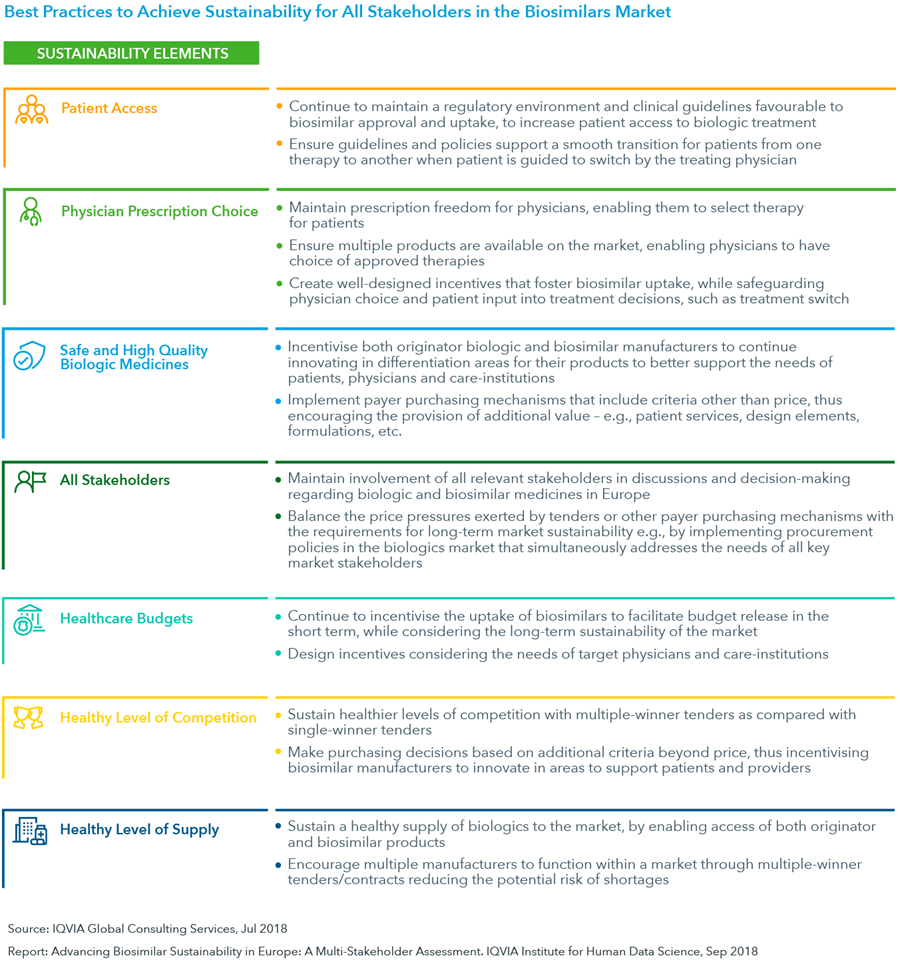
- Based on sustainable policies currently in place in Europe, a set of best practices can be leveraged to ensure long-term sustainability of the biosimilars market, while additionally fulfilling the needs of all stakeholders.
- These include creating positive incentives to promote biosimilar uptake, taking steps to ensure that physicians retain prescription freedom to offer the best product selection for a specific patient and designing tenders and contracts to have multiple winners and include criteria other than price.
















