Specialized expertise and customized solutions across 14 therapeutic centers of excellence, including oncology, GI/NASH, pediatrics, neurology and rare diseases.
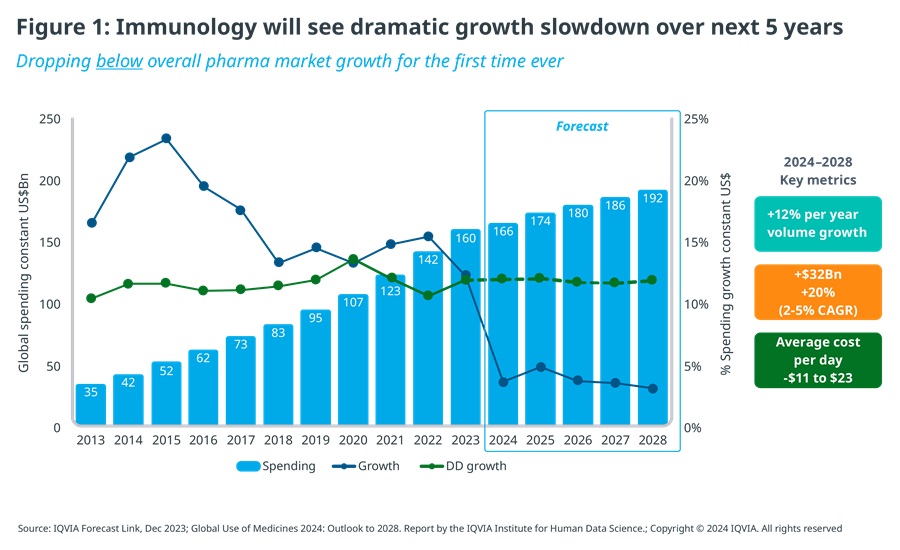
For the past two decades, immunology has been one of the major growth engines of the biopharmaceutical industry, delivering sustained, double-digit growth ranging from 12% to 23% YoY, about 3-5 times the rate of the overall pharmaceutical market for prescription medicines. By the end of 2024, it is forecast to reach $166 billion globally, at ex-manufacturer prices, which makes immunology the second largest therapy area by value, behind oncology.
Slowing growth and overcrowding
The outlook for immunology for the next five years, however, is a very different one. Despite ongoing innovation and the continuing increase in the number of treated patients, value growth is expected to slow down dramatically, from 15% CAGR for the past 5 years to 2-5% CAGR for the 5 years ahead – dropping, for the first time ever, below overall pharmaceutical market growth.
This is the result of biosimilars of first-generation autoimmune blockbusters, especially Humira and Stelara which are widely used across multiple indications, eroding top-line growth by exerting downward price pressure on originators or substituting originator volume with lower cost alternatives (see Figure 1).
In addition to the impact of first-generation blockbuster LoEs, all major immunology indications are ‘over-heating’, as frantic innovation activity is leading to extensive crowding and fierce competition between and within drug classes.
As a result of current mid- to late-stage pipeline assets being launched, over the next 3-5 years all major autoimmune indications, including rheumatoid arthritis, psoriasis, psoriatic arthritis, Crohn’s disease and ulcerative colitis, are likely to see at least 6-7 MoAs and 12-15 brands compete in each indication. This rises to >10 MoAs and >20 brands per indication in the most competitive scenarios, excluding additional competitive pressure from biosimilars of relevant therapies, such as infliximab, etanercept, adalimumab or ustekinumab. Among inflammation indications, atopic dermatitis is heading this way too (see Figure 2).
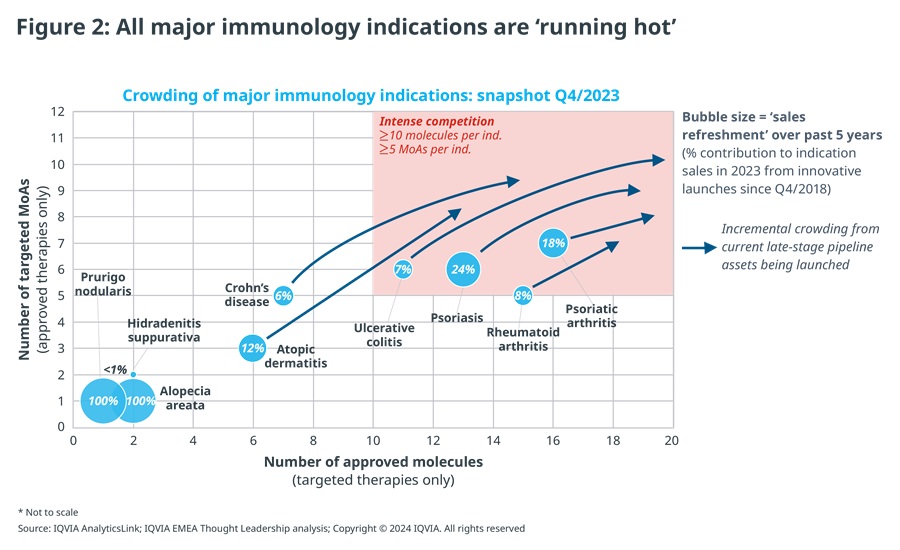
Despite such frantic levels of innovation, sales refreshment – the contribution to indication sales from new products launched in the past 5 years – is weighed down by deeply entrenched, first-generation therapies such as various anti-TNF inhibitors, Stelara and respective biosimilars of those therapies. Furthermore, a fragmented innovation frontier, with multiple new products jostling for position, gives payers choice and in turn more leverage to extract value, while limiting opportunities for new launches and the possibility of a future mega-blockbuster.v
Consequently, sales refreshment failed to reach 25% among major immunology indications, even for those seeing the most intense innovation activity, such as psoriasis, where multiple recent launches introduced new, differentiated therapy options with both established and new MoAs, e.g., IL-17, IL-23 and TYK-2. Sales refreshment was even lower in all other major indications.
Seeking new horizons
Unsurprisingly, innovators have been compelled to look elsewhere. In pursuit of future growth, they are increasingly looking towards smaller immunology indications for untapped opportunities and the generally less mature inflammation segment within immunology, which still offers ample white space (see Figure 3).
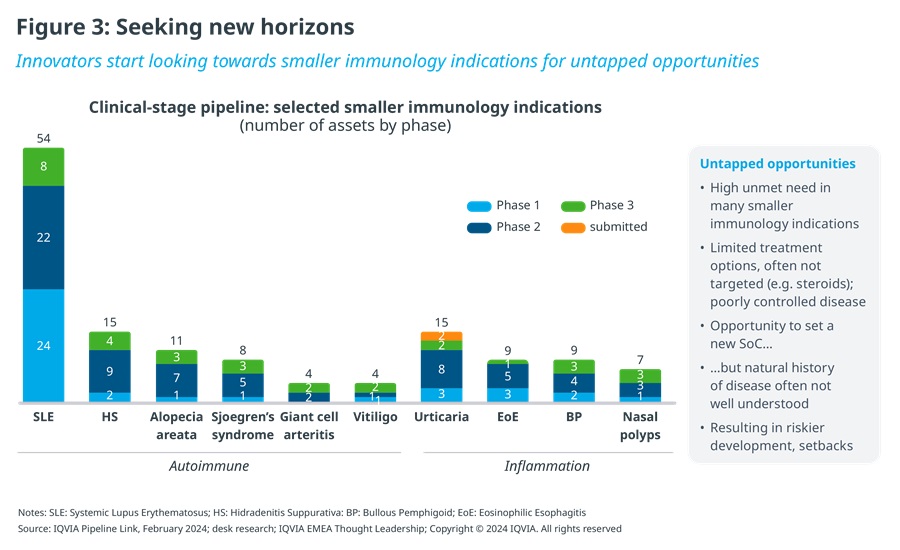
Unmet need is high in a long list of largely neglected immunological diseases, such as systemic lupus erythematosus, hidradenitis suppurativa, alopecia areata, Sjögren's syndrome, vitiligo, bullous pemphigoid or prurigo nodularis. Their prevalence spans a range from rare diseases with fewer than 200 thousand patients in the US, for example, to relatively more common conditions with a million or more patients, a subset of whom are severe cases most in need of innovative therapies. What they share is having been underserved, with typically limited disease-specific treatment options available to date, leaving many patients with poorly controlled disease. This represents an attractive opportunity for innovators to set an effective, new standard of care.
However, the natural history of these diseases is often less well understood and has led to several setbacks, e.g., in lupus. Nevertheless, innovators are clearly not disheartened, judging by recent pipeline momentum. For example, hidradenitis suppurativa has seen a major inflection point in development activity after many years of an innovation drought and was a key focus at the recent annual meeting of the American Academy of Dermatology (AAD), with several high-profile presentations of mid- to late-stage readouts, including AbbVie’s IL-1α/1β antagonist lutikizumab, Novartis’ oral BTK inhibitor remibrutinib, and Moonlake’s anti-IL-17 nanobody solenokimab.
The overall change in innovation intensity that we are witnessing across underserved immunology diseases is reminiscent of the momentum that transformed today’s major autoimmune indications about 10-15 years ago. However, without such epidemiological scale, innovators today must play across multiple of those smaller indications to achieve critical mass in an immunology franchise, e.g., via a portfolio of assets and/or multi-indication assets.
Seizing the opportunity
Finding success in less explored immunology indications requires a different approach. Unlike the major, well-established autoimmune conditions such as RA, psoriasis or Crohn’s disease, smaller indications face unique challenges, for example, often low disease awareness among patients and HCPs, a limited understanding of the burden of illness and its true impact on patients’ life, immature care pathways, including diagnosis, specialist referral and treatment, leading to under-diagnosis and under-treatment, or convincing payers of the need to treat and for them to cover novel therapies.
Innovators targeting smaller, oft neglected immunology indications therefore must focus on three priorities:
- Patient-relevant evidence, going beyond traditional endpoints used in immunology to assess true patient impact, including QoL. For example, we have seen recent efforts in defining clinically more meaningful and patient-relevant composite endpoints for Sjögren's syndrome, e.g., CRESS (Composite of Relevant Endpoints for Sjögren's Syndrome), combining 5 complementary measures that collectively better capture disease severity to assess treatment response and patient outcomes, given the heterogeneous and complex nature of primary Sjögren's syndrome. Partnering with medical societies and opinion leaders is critical for validation and acceptance of novel endpoints.
- Close collaboration with patient advocacy groups, for example to gain input from patients in defining patient-relevant endpoints, help with patient recruitment for clinical trials, support disease awareness building or as patient advocates in efforts to secure market access and reimbursement for novel therapies.
- Extensive market shaping and health system preparation, focussing on raising awareness of the disease, unmet need and its patient burden; building advocacy for the need to treat with innovative therapies and shaping treatment guidelines; and facilitating health system readiness, e.g., establishing or streamlining care pathways, or embedding new diagnostic tools and standards. This requires early, external engagement, especially medical affairs-led, and the deployment of dedicated roles focussed on health system enablement.
As innovators re-direct their efforts towards historically underserved immunological diseases, long-suffering patients will be the ultimate winners, as the prospect of effective treatment options makes big strides towards becoming reality.
















