Create exceptional customer experiences through intelligence-driven personal engagements.

Rethink Engagement. Redefine Value.
More stakeholders, more channels and higher expectations require life science companies to take a smarter approach to customer engagement. Meet HCPs on their terms with seamless, personalized engagements that fulfills changing needs and improves results.
IQVIA OCE bridges intelligence with experience for HCPs and commercial teams alike. Intelligent connections across the commercial ecosystem improve collaboration across teams, drives HCP-centric experiences, and enables user-centric smart workflows for better decision making.

Renovate your Sales Model. Upgrade Customer Outcomes
Watch this demo video and learn how OCE Personal, an integrated CRM, seamlessly incorporates actionable insights directly into the user interface. By modernizing the sales model and enabling full collaboration across team members, OCE Personal empowers sellers with smarter and more effective customer engagements, allowing organizations to optimize sales models, elevate customer outcomes, and reinvent HCP engagement.
Work smarter, not harder
OCE+ adds IQVIA’s Next Best Recommendation Engine to our industry-leading Orchestrated Customer Engagement (OCE) platform. Embedded intelligence provides insights in daily workflows to make taking action easy.
The result. Better HCP experiences, improved productivity, and increased ROI for life sciences companies.

IQVIA, Alibaba & Salesforce: The Future of Life Sciences in China

Intelligence-powered commercial teams

2022 Gartner® Market Guide for CRM in Life Sciences
IQVIA’s Orchestrated Customer Engagement (OCE), built on Salesforce, enables more personalized, compliant interactions between life science companies and HCPs by leveraging embedded intelligence to drive actionable insights.
IQVIA is proud to be mentioned as a representative vendor in the 2022 Gartner Market Guide for CRM – read the guide to learn more.

IQVIA named a Leader by Everest Group
The 2023 Everest Group’s Life Science Customer Experience Platforms PEAK Matrix® assessment evaluated products from 18 companies serving the industry. IQVIA’s OCE platform was one of only three to earn honors as a Leader.
Please click below to access the custom report.
IQVIA ORCHESTRATED CUSTOMER ENGAGEMENT SOLUTIONS
Coordinate multichannel marketing touch points for timely and personalized communications.
Empower HCPs with on-demand access to digital services and compliant, personalized resources.
Optimize resources and engagement plans across all customer interactions.
Enable compliant planning, management, contracting & payment of HCPs & HCOs globally.
Engage remotely on your customer’s terms in a flexible, personalized, compliant manner.
An expansion of the OCE platform to meet the evolving needs of life sciences commercial teams, OCE+ enables a smarter way to work.
Seamlessly customize instances of OCE with our platform for easy app build and integration

ON-DEMAND WEBINAR
IQVIA OCE July 2024 Release Webinar
Join us for our July OCE product release webinar to learn more about key themes, new innovations and high-level roadmap for the OCE platform.

CUSTOMER SUCCESS STORY
OCE+ improves engagement to drive sales growth
See how a midsize global life science company optimized field force execution by adopting IQVIA OCE+ to improve HCP engagement and drive sales growth. With OCE+, the company could leverage a CRM built to contextually deliver AI/ML-driven insights to the field team to optimally engage customers both in-person and through digital channels – better positioning the commercial team for improved productivity and increased ROI.
The client realized an immediate sales uplift shortly after deploying, driving higher sales growth with adoption over 80% by week four. Download now to read more.

We required an engagement solution that allowed our sales teams to provide a high-level of personalized interactions with our healthcare providers. IQVIA’s OCE+ allows them to adapt in real-time to the changing need of our customers.
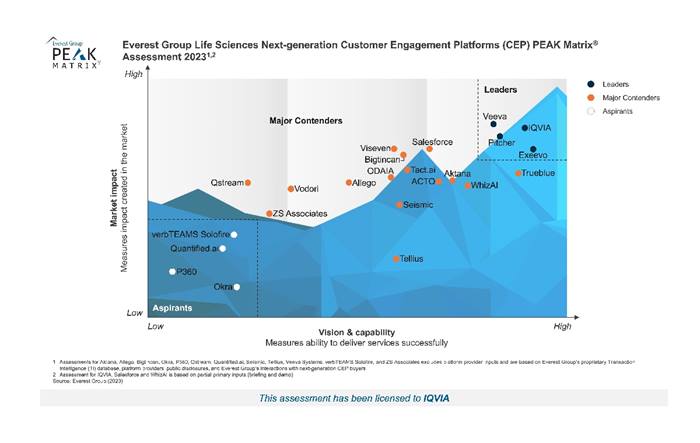
IQVIA recognized as Leader for Next-Generation Customer Engagement Platform
IQVIA has been named a ‘Leader’ in the Everest Group Life Sciences Next-Generation Customer Engagement Platforms PEAK Matrix® Assessment 2023.
This is the third 2023 PEAK Matrix Assessment in which IQVIA has been positioned high in the “leader category.
Please click below to access more details.

Blog
Comprehensive customer engagement channel optimization functionalities
Check out this blog post to see why, out of the 23 leading life sciences customer engagement platform providers, IQVIA’s Orchestrated Customer Engagement suite was identified as a member of the “Leader” category in the Everest Group Life Sciences Next-Generation Customer Engagement Platforms PEAK Matrix® Assessment 2023.
IQVIA continues to be recognized as industry leader
The 2023 Everest Group Life Sciences Operations PEAK Matrix® Assessment evaluated products from 28 companies serving the industry.
IQVIA was named a ‘Leader’ and positioned highest of all companies surveyed.
Click below to access the custom report.
The 2023 Everest Group’s Life Sciences Sales and Marketing Operations PEAK Matrix® Assessment evaluated 17 life sciences sales and marketing operations suppliers servicing the industry.
IQVIA was the highest-scoring provider named in the Leader category.
Click below to access the full report for more details.

Understanding Implications to Your CRM Environment
Many life sciences organizations are undergoing a multiyear journey to transform their customer engagement ecosystems and enable digital-first, content-rich experiences.
Check out this blog to evaluate your current state and get recommendations from Gartner to navigate your company’s CRM transition.


























