





















- Insights
- The IQVIA Institute
- Reports and Publications
- Reports
- Upholding the Clinical Promise of Precision Medicine
About the Report
An understanding of systems biology has allowed for greater insight into the processes that govern disease and has helped to usher in the era of precision medicine—where the right patient gets the right therapy at the right dose and experiences the right response. This report provides key insights into the characterization of systems therapeutics and precision medicines. It also reports on their use and spending though 2016. The report introduces a framework for understanding the unique challenges associated with these therapies including cost, clinical decision making and the roles of healthcare stakeholders.
Summary
Recent advances in genomics, imaging, and biomarkers are translating into the development and use of systems therapeutics, which are medicines that target specific pathways to modify the course of disease and are intended for personalized or stratified use in specific patient populations. Precision medicines are a subset within systems therapeutics and have seen a period of rapid growth and acceptance within the healthcare system, bringing the benefit of more effective treatments to patients along with reduced adverse effects. This paradigm shift in medicine will have resounding impacts on all aspects of the healthcare system.
Medicines included in the analysis in this report came with a recommendation or requirement on their label or were based on regulatory or professional-society testing for a specific gene, protein, hormone or viral genotyping. Of these medicines, 58% were from the oncology therapy area, 69% received regulatory approval after 2006, and 80% were from the small molecule drug class. 4.5 billion standard units were used by patients in the ten major developed markets in 2016 out of 4 trillion units for global medicines, with significant use among older, small molecule CNS and breast cancer therapies. Total spending on stratified medicines in the 10 developed markets totaled over $62.8Bn in 2016 and was heavily influenced by oncology and antiviral therapies. The report notes that identification of patients with predictive biomarkers is the most crucial step in the current framework for proper use of precision medicines and this first step is influenced by cost, access to diagnostics as well as physician and payer acceptance. Other challenges identified in the optimal use of precision medicines include geographic availability, appropriate use and pharmacovigilance.
Key Findings
The Number of Precision Medicines Has Increased Steadily Since 1977
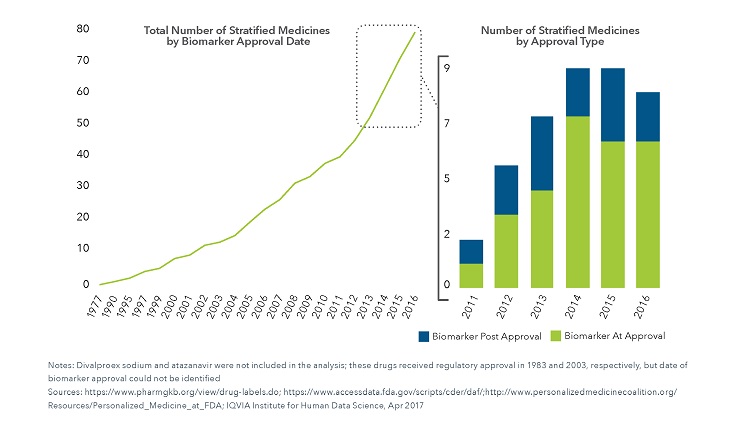
- Stratified medicines included in the analysis in this report came with a recommendation or requirement on their label or were based on regulatory or professional society testing of a specific gene, protein, or hormone in either the United States, Canada, Japan or Europe. Products where testing of viral genotype was required prior to use were also included within the analysis.
- 80 unique therapies were included in the analysis based on the inclusion of pharmacogenomic or other biomarker testing requirements in their label. 69% of the therapies were approved from 2006-2016. Although the number of therapies that stratify patients according to biomarkers has accumulated steadily since 1977, the trend for new precision medicines has begun to stabilize.
- 21 therapies received regulatory approval for a pharmacogenomic biomarker that directed patient stratification after the initial market approval, likely due to advances in understanding of disease as well as added costs of research and development for biomarker stratification.
4.5 Billion Standard Units for Stratified Medicines Were Used by Patients in the Ten Major Developed Markets in 2016
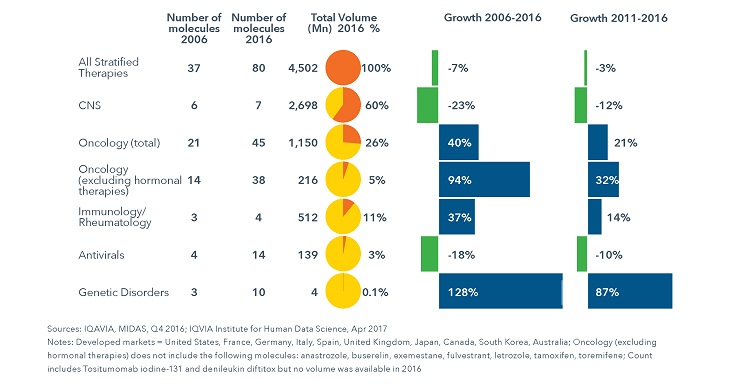
- Total use of stratified medicines in the developed markets, measured in standard units, has remained relatively flat since 2006. This is primarily due to the higher representation of small molecule therapies within the group, many of which experienced generic competition within the study period, balanced against the launch of novel oncology and antiviral therapies with low volumes.
- Stratified oncology medicines accounted for 26% of use and saw a growth of 40% from 2006-2016.
- Within stratified medicines, older small molecules, particularly those in CNS and breast cancer, account for over 75% of the volume.
Stratification of Patient Populations with Predictive Biomarkers is the First Step in Use of Precision Medicines and Can Present a Bottle Neck in Terms of Access and Reimbursement
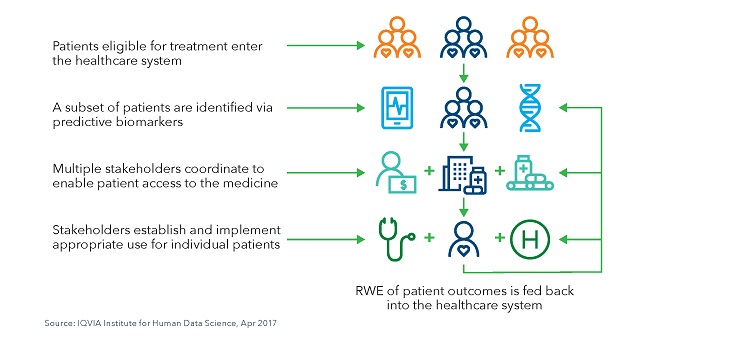
- The use of predictive biomarkers is the first critical step for the identification of patients for precision medicines and a biomarker is considered predictive should the effect of treatment show a difference between those patients screened positive for a biomarker compared with those who show a negative screen.
- Optimal use of precision medicines will depend in part on patient access. Importantly, timely access is critical, and coordination among stakeholders is required to move smoothly from companion diagnostic testing and interpretation of results to efficient access to precision medicines.
- The use of stratified oncology medicines is particularly complex. Predictive biomarkers are essential for treatment in many oncology areas, and a personalized protocol for each individual patient is becoming the norm. This increased complexity can lead to difficult treatment decisions physicians who must draw from clinical experience, disease area guidelines, academic research and payer prescription guidelines.
- Analyzing patient outcomes helps to provide essential information on the use of systems therapeutics that can be used to influence future treatments. However, the volume of data from patients receiving systems therapeutics is often larger, and more complex, than for traditional medicines. These large data sets will need to be effectively protected and shared between stakeholders to provide the feedback necessary to support the use of these therapies in future patients.





