Build on our experience of more than 245 rare disease studies in 96 countries to fulfill your promise of hope to millions around the world.
-
Americas
-
Asia & Oceania
-
A-I
J-Z

EMEA Thought Leadership
Developing IQVIA’s positions on key trends in the pharma and life sciences industries, with a focus on EMEA.
Learn more -
Middle East & Africa

EMEA Thought Leadership
Developing IQVIA’s positions on key trends in the pharma and life sciences industries, with a focus on EMEA.
Learn more
Regions
-
Americas
-
Asia & Oceania
-
Europe
-
Middle East & Africa
-
Americas
-
Asia & Oceania
-
Europe
Europe
- Adriatic
- Belgium
- Bulgaria
- Czech Republic
- Deutschland
- España
- France
- Greece
- Hungary
- Ireland
- Israel
- Italia

EMEA Thought Leadership
Developing IQVIA’s positions on key trends in the pharma and life sciences industries, with a focus on EMEA.
Learn more -
Middle East & Africa

EMEA Thought Leadership
Developing IQVIA’s positions on key trends in the pharma and life sciences industries, with a focus on EMEA.
Learn more
SOLUTIONS
-
Research & Development
-
Real World Evidence
-
Commercialization
-
Safety & Regulatory Compliance
-
Technologies
LIFE SCIENCE SEGMENTS
HEALTHCARE SEGMENTS
- Information Partner Services
- Financial Institutions
- Public Health and Government
- Patient Associations
- Payers
- Providers
THERAPEUTIC AREAS
- Cardiovascular
- Cell and Gene Therapy
- Central Nervous System
- GI & Hepatology
- Infectious Diseases and Vaccines
- Oncology
- Pediatrics
- Rare Diseases
- View All

Impacting People's Lives
"We strive to help improve outcomes and create a healthier, more sustainable world for people everywhere.
LEARN MORE
Harness the power to transform clinical development
Reimagine clinical development by intelligently connecting data, technology, and analytics to optimize your trials. The result? Faster decision making and reduced risk so you can deliver life-changing therapies faster.
Research & Development OverviewResearch & Development Quick Links

Real World Evidence. Real Confidence. Real Results.
Generate and disseminate evidence that answers crucial clinical, regulatory and commercial questions, enabling you to drive smarter decisions and meet your stakeholder needs with confidence.
REAL WORLD EVIDENCE OVERVIEWReal World Evidence Quick Links

See markets more clearly. Opportunities more often.
Elevate commercial models with precision and speed using AI-driven analytics and technology that illuminate hidden insights in data.
COMMERCIALIZATION OVERVIEWCommercialization Quick Links

Service driven. Tech-enabled. Integrated compliance.
Orchestrate your success across the complete compliance lifecycle with best-in-class services and solutions for safety, regulatory, quality and medical information.
COMPLIANCE OVERVIEWSafety & Regulatory Compliance Quick Links

Intelligence that transforms life sciences end-to-end.
When your destination is a healthier world, making intelligent connections between data, technology, and services is your roadmap.
TECHNOLOGIES OVERVIEWTechnology Quick Links
CLINICAL PRODUCTS
COMMERCIAL PRODUCTS
COMPLIANCE, SAFETY, REG PRODUCTS
BLOGS, WHITE PAPERS & CASE STUDIES
Explore our library of insights, thought leadership, and the latest topics & trends in healthcare.
DISCOVER INSIGHTSTHE IQVIA INSTITUTE
An in-depth exploration of the global healthcare ecosystem with timely research, insightful analysis, and scientific expertise.
SEE LATEST REPORTSFEATURED INNOVATIONS
-
IQVIA Connected Intelligence™
-
IQVIA Healthcare-grade AI™
-
Human Data Science Cloud
-
IQVIA Innovation Hub
-
Decentralized Trials
-
Patient Experience powered by Apple
WHO WE ARE
- Our Story
- Our Impact
- Commitment to Public Health
- Code of Conduct
- Environmental Social Governance
- Privacy
- Executive Team
NEWS & RESOURCES

Unlock your potential to drive healthcare forward
By making intelligent connections between your needs, our capabilities, and the healthcare ecosystem, we can help you be more agile, accelerate results, and improve patient outcomes.
LEARN MORE
IQVIA AI is Healthcare-grade AI
Building on a rich history of developing AI for healthcare, IQVIA AI connects the right data, technology, and expertise to address the unique needs of healthcare. It's what we call Healthcare-grade AI.
LEARN MORE
Your healthcare data deserves more than just a cloud.
The IQVIA Human Data Science Cloud is our unique capability designed to enable healthcare-grade analytics, tools, and data management solutions to deliver fit-for-purpose global data at scale.
LEARN MORE
Innovations make an impact when bold ideas meet powerful partnerships
The IQVIA Innovation Hub connects start-ups with the extensive IQVIA network of assets, resources, clients, and partners. Together, we can help lead the future of healthcare with the extensive IQVIA network of assets, resources, clients, and partners.
LEARN MORE
Proven, faster DCT solutions
IQVIA Decentralized Trials deliver purpose-built clinical services and technologies that engage the right patients wherever they are. Our hybrid and fully virtual solutions have been used more than any others.
LEARN MORE
IQVIA Patient Experience Solutions powered by Apple
Empowering patients to personalize their healthcare and connecting them to caregivers has the potential to change the care delivery paradigm. IQVIA and Apple are collaborating to bring this exciting future of personalized care directly to devices patients already have and use.
LEARN MOREWORKING AT IQVIA
Our mission is to accelerate innovation for a healthier world. Together, we can solve customer challenges and improve patient lives.
LEARN MORELIFE AT IQVIA
Careers, culture and everything in between. Find out what’s going on right here, right now.
LEARN MORE
WE’RE HIRING
"Improving human health requires brave thinkers who are willing to explore new ideas and build on successes. Unleash your potential with us.
SEARCH JOBSWith new legislative initiatives from regulatory bodies in a number of healthcare markets, coupled with a high-unmet medical need, pharmaceutical companies are continuing to invest in the development of orphan drugs. This article provides an analysis of key 2019 orphan drug approvals, using IQVIA Pipeline Intelligence data.
Introduction
Rare diseases, for which orphan drugs are indicated to treat, are characterized by a low frequency of occurrence in the general population of a geographical region. There are over 8000 described rare diseases, of which only 340 conditions currently have treatment options1. Some of these diseases, such as progeria, occur in 1 in every 4 million births, while others may be comparatively more common, such as multiple sclerosis, cystic fibrosis and Duchenne muscular dystrophy. The prevalence of these diseases varies between populations, making an indication rare for one region but common for another. The effects of these rare diseases range from premature mortality to severe disability due to a lack of efficacious treatment options. Growing understanding of numerous aspects of these diseases, along with improving government policies, are expanding the framework for research, development and market penetration through the introduction of novel and renovated therapies.
2019 Orphan Drug Approval Overview
The improvement in government policies over the years has been mirrored by an increase in the development activities for candidates addressing rare diseases; with less than 10 approved rare disease therapies on the market in the 1970s increasing to more than 830 US FDA approved orphan therapies as of 20192. New treatment options have been brought to market through the launch of either new molecular entities or through the renovation of older, established therapies.
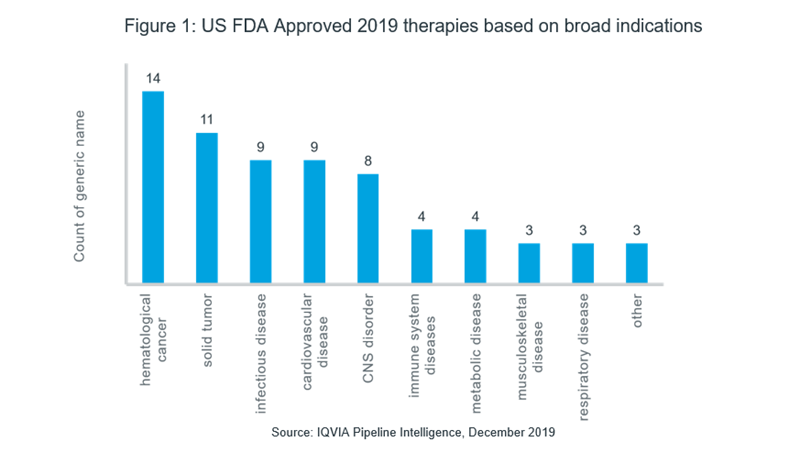
As shown in Figure 1, the US FDA granted approval to more than 65 therapies2 in various rare disease therapeutic areas with the most approvals seen in cancer. Among them, hematological cancers have seen the maximum approvals, with 14 therapies for a range of indications including; multiple myeloma, myelofibrosis, acute lymphoid leukemia and various forms of lymphomas approved in 2019. This is followed by 11 registered therapies for solid tumors, including small cell lung cancer, non-small cell lung cancer, ovarian cancer, hepatocellular cancer, esophageal cancer, neuroendocrine tumor, melanoma and NTRK fusion-positive tumors. Furthermore, 9 therapies were approved for infectious diseases, including 2 therapies for tuberculosis, 3 for hepatitis C virus infection, 2 for human immunodeficiency virus infection and a therapy for fascioliasis. With 8 therapy approvals in the USA for disorders of the central nervous system, including Lambert-Eaton myasthenic syndrome, neuromyelitis optica, spinal muscular atrophy, amyotrophic lateral sclerosis and narcolepsy, the needs of another major therapeutic area have to some extent, been catered to. Additionally, 2 biologic therapies have been approved for Churg-Strauss Syndrome, 1 for graft versus host disease and 1 for systemic sclerosis in the field of autoimmune diseases.
Conclusion
With increasing focus from governments and regulatory bodies, coupled with a high-unmet medical need, pharmaceutical companies will continue to invest in the development of orphan drugs. In particular, ‘Big Pharma’ will continue to explore this field, through its own organic development and through the acquisition of small start-ups as well as established players. Additionally, the recent launch of a one-time adeno-associated virus mediated human RPE65 gene therapy for Leber congenital amaurosis, clearly establishes the will of big pharma to explore the orphan drug space, despite the financial and research headwinds.
References
- Ekins, S. Industrializing rare disease therapy discovery and development. Nature Biotechnology 2017; 35(2), 117–118.
- US FDA Orphan Drug list access portal. Available at: http://bit.ly/34xkdDg
- Melnikova, I. Rare diseases and orphan drugs. Nature Reviews Drug Discovery, 2012;11(4), 267–268.
To find out more about IQVIA Pipeline Intelligence is, please visit
https://www.iqvia.com/solutions/commercialization/brand-strategy-and-management/product-pipeline
You may also be interested in
Related solutions
Specialized expertise and customized solutions across 14 therapeutic centers of excellence, including oncology, GI/NASH, pediatrics, neurology and rare diseases.











