





















- Blogs
- Securing market access with single-arm trial data: Why external comparators could be the answer
HTA submissions of single-arm evidence appear to fare better when they include an external comparator. However, with many factors affecting HTA decision-making, detailed analysis and a clear strategy are essential to determine the most appropriate approach and external comparator for your target indication.
R&D pipelines are rich with innovation in areas of high unmet need, driving a surge in promising new treatments for patients with niche and rare diseases. Under pressure to accelerate approval, regulators are increasingly authorizing use on the basis of single-arm efficacy data. But how can you secure recommendation for HTA reimbursement? Do external comparators make a difference? We’ve analyzed the evidence from IQVIA’s HTA Accelerator database to find out.
HTA Accelerator provides instant insights into payer decision-making from HTA reports of more than 100 agencies in 32 countries across all therapeutic areas. Based on our data extraction parameters1 we’ve identified 433 single-arm clinical trial submissions from 2011 to 2019 with a year-on-year increase, mostly in oncology indications (see Figure 1). Overall, 48% (208/433) have received a positive recommendation.
Figure 1: Single-arm Clinical Trial submissions to HTA bodies (Jan 2011 to Dec 2019)

Use of external comparators
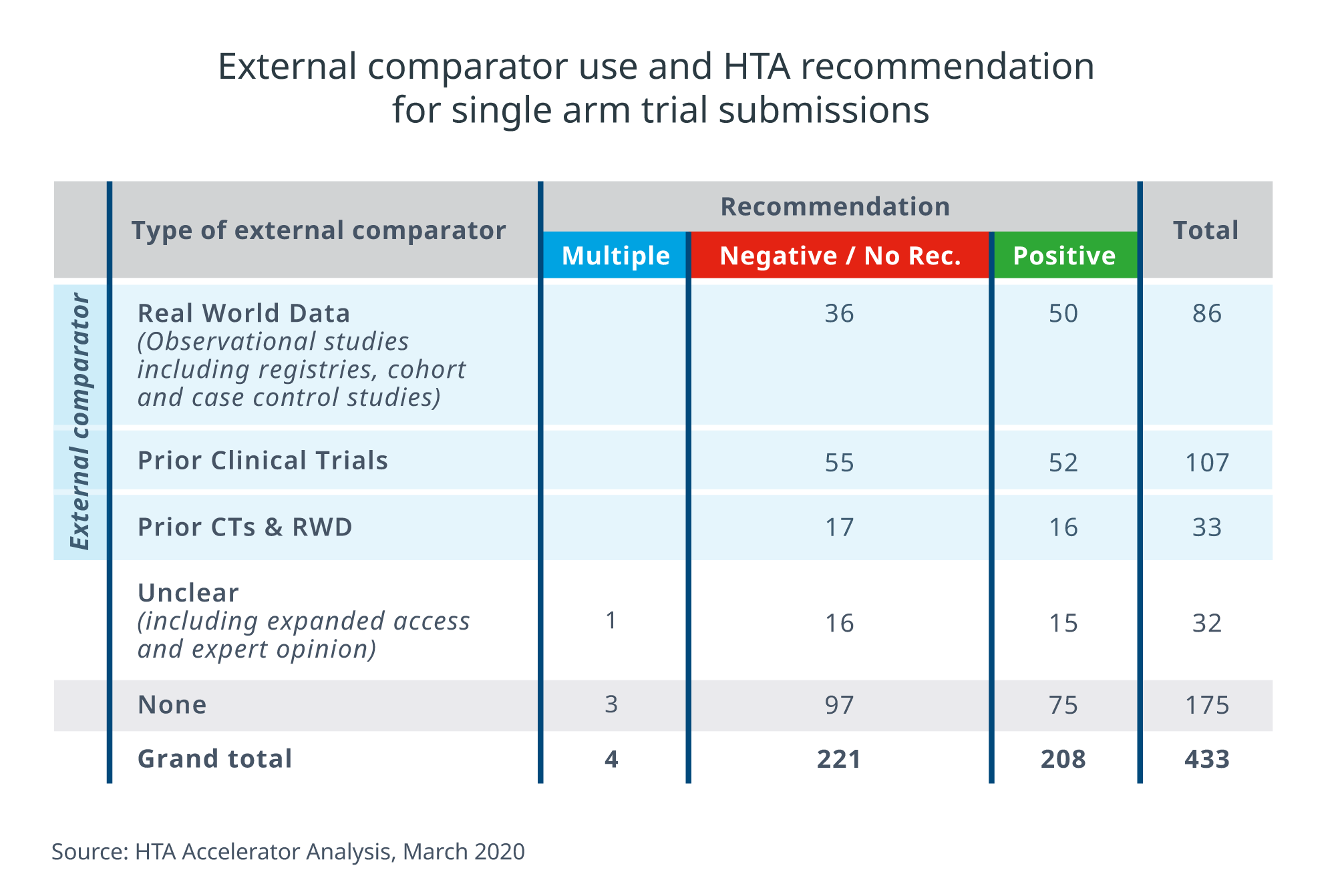
In 52% (226/433) of cases, manufacturers clearly used some kind of external comparator to demonstrate clinical and economic benefit. External comparators are patient cohorts from outside the trial. Generated from other trials or retrospective/observational real world data, they are used to make indirect comparisons with the trial cohort or to serve as real-world benchmarks. Our analysis shows that a variety of external comparator datasets were used in HTA packages, the most common being prior RCT data (47%, 107/226) or some type of real world study data (38%, 86/226) (see Figure 2).
Figure 2: Single-arm HTA submission outcome by type of external comparator used
Better results but many factors to consider
We see a trend towards better results when using external comparators. Overall, 52% (118/226) of external comparator submissions gained a positive HTA recommendation compared to 43% (75/175) of HTA submissions with no external comparator data. However, the presence of an external comparator does not guarantee HTA acceptance. Based on our analysis of post-HTA conclusions, acceptability of the data and appropriateness of the analytical method were important considerations, underscoring the multidimensional nature of HTA decision-making. How, then, can you decide which way to go?
A four-step strategic process
There is no formal guidance or established best practice for tackling HTA submissions with single-arm evidence. Choosing comparator cohorts isn’t easy. Standards of patient care can change over time and vary by country. Careful evaluation of past HTA decisions in your target indication(s) as well as early input from HTA stakeholders are critical. Before designing an external comparator study, we recommend a strategic approach to
- Establish the need for an external comparator by anticipating the strength of your clinical trial and evaluating the competitive landscape
- Select the right external comparator group to match your trial population, appreciating the clinical and statistical implications of any differences
- Determine the optimal approach to data sourcing based on the available options and your budget, time and geographic restrictions
- Identify the best methodology for comparison to your trial data, understanding the relative suitability of different indirect comparison approaches versus use of a real world benchmark
IQVIA has a dedicated team of experts with significant experience working with manufacturers to define the strategy and methodology for external comparators supporting submissions to regulators and HTA bodies.
For further information contact Joss.Warren@iqvia.com or Dony.Patel@iqvia.com
[1] Inclusion criteria = Single-arm study; Original submission or extension of indication; All countries; All years 1996–Sept 2018.





